 The opening and closing repeats continuously in an ongoing interconversion between anomeric forms and is referred to as mutarotation (Latin mutare, meaning “to change”). You can start with a pure crystalline sample of glucose consisting entirely of either anomer, but as soon as the molecules dissolve in water, they open to form the carbonyl group and then re-close to form either the α or the β anomer. When the sample is dissolved in water, however, a mixture is soon produced containing both anomers as well as the straight-chain form, in dynamic equilibrium. The α form melts at 146☌ and has a specific rotation of +112°, while the β form melts at 150☌ and has a specific rotation of +18.7°. It is possible to obtain a sample of crystalline glucose in which all the molecules have the α structure or all have the β structure. The beta anomer is preferred because β -D-glucopyranose is the only aldohexose which can be drawn with all its bulky substituents (-OH and -CH 2OH) in equatorial positions, making it the most stable of the eight D-aldohexoses, which probably accounts for its widespread prevalence in nature. When D-glucose cyclizes it forms a 37/63 mixture of the alpha and beta anomer respectively. Also, the terminal -CH 2OH group is oriented up on the pyranose ring for D-sugars and down for L-Sugars. Groups which go to the right in a Fischer projection will be orientend 'down' of the pyranose ring while groups to the left are oriented 'up' in the chair structure. By convention the ring oxygen is placed to the right and to the rear of the structure (top right of the drawing). Pyranose rings are even capable of undergoing a ring flip to change between chair conformations. Pyranose rings are often drawn in a chair conformation like cyclohexane rings ( Section 4-6) with substituents being either an axial or equatorial position. Notice that for glucose and other aldohexoses the hydroxyl that forms the cyclic hemiacetal is also the one that determines the D/L designation of a sugar. The cyclic form of glucose is called glucopyranose. The pyranose ring is formed by attack of the hydroxyl on carbon 5 of glucose to the aldehyde carbon (carbon #1, also called the anomeric carbon in carbohydrate terminology). At equilibrium less than 1% of glucose is in an open chain form with the rest being almost exclusively in its cyclic pyranose form. The size of the cyclic hemiacetal ring adopted by a given sugar is not constant, but may vary with substituents and other structural features.Īldohexoses usually form pyranose rings and their pentose homologs tend to prefer the furanose form, but there are many counter examples. For most five- and six-carbon sugars, the cyclic forms predominate in equilibrium since they are more stable. Sugars are often shown in their open-chain form, however, in aqueous solution, glucose, fructose, and other sugars of five or six carbons rapidly interconvert between straight-chain and cyclic forms. Unlike most of the biochemical reactions you will see in this text, sugar cyclization reactions are not catalyzed by enzymes: they occur spontaneously and reversibly in aqueous solution. 
Cyclic structures of this kind are termed furanose (five-membered) or pyranose (six-membered), reflecting the ring size relationship to the common heterocyclic compounds furan and pyran shown below.įuran (5-membered ring) and pyran (6-membered ring) structures Five and six-membered rings are favored over other ring sizes because of their low angle and eclipsing strain. 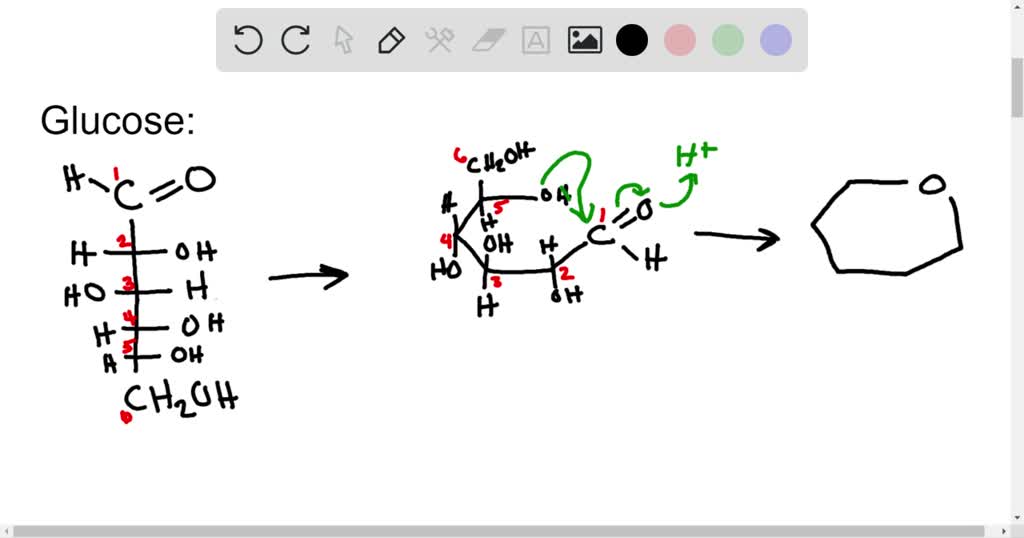
Molecules which have both an alcohol and a carbonyl can undergo an intramolecular version of the same reaction forming a cyclic hemiacetal.īecause sugars often contain alcohol and carbonyl functional groups, intramolecular hemiacetal formation is common in carbohydrate chemistry. In Section 19-10 it was discussed that the reaction of one equivalent of an alcohol, in the presence of an acid catalyst, adds reversibly to aldehydes and ketones to form a hydroxy ether called a hemiacetal (R 2COHOR') (h emi, Greek, half). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
